|
This comprehensive perspective not only helps students grasp fundamental concepts but also encourages critical thinking and analytical skills development.įurthermore, a reference table serves as a reliable guide for students to adhere to proper scientific conventions. By exploring the various sections and elements within the table, students can develop a holistic understanding of patterns, trends, and relationships between different elements. Beyond its function as a mere repository of data, it serves as a valuable educational aid. Moreover, a reference table promotes a deeper understanding of chemistry concepts. Students can swiftly cross-reference values, verify equations, and ensure accurate results, thus streamlining their workflow and allowing for more productive study sessions. In such situations, referring to a well-structured reference table can significantly speed up the process. With the complex nature of chemistry, students often find themselves grappling with intricate calculations, conversions, or the need to identify properties of different elements. why we use Reference table?Īdditionally, a reference table fosters efficiency and saves valuable time. By having all these resources conveniently organized in one place, students can easily locate and retrieve the necessary information when working on assignments, conducting experiments, or preparing for exams. This compilation encompasses a wide range of chemical elements, compounds, equations, and other essential information that students encounter in their studies. To begin with, a reference table acts as a comprehensive repository of key data and formulas. It facilitates access to relevant information and aids in learning and academic performance. It provides a quick and easy reference for key concepts and principles in chemistry.Ī reference table is useful for everybody, regardless of their level of chemical expertise. Chemistry Reference Table IntroductionĪ chemistry reference table is a compilation of essential information and data for use by students and professionals. A Chemistry Reference Table is an essential resource for every chemistry student. To succeed as a scientist in school, you need access to the appropriate materials. 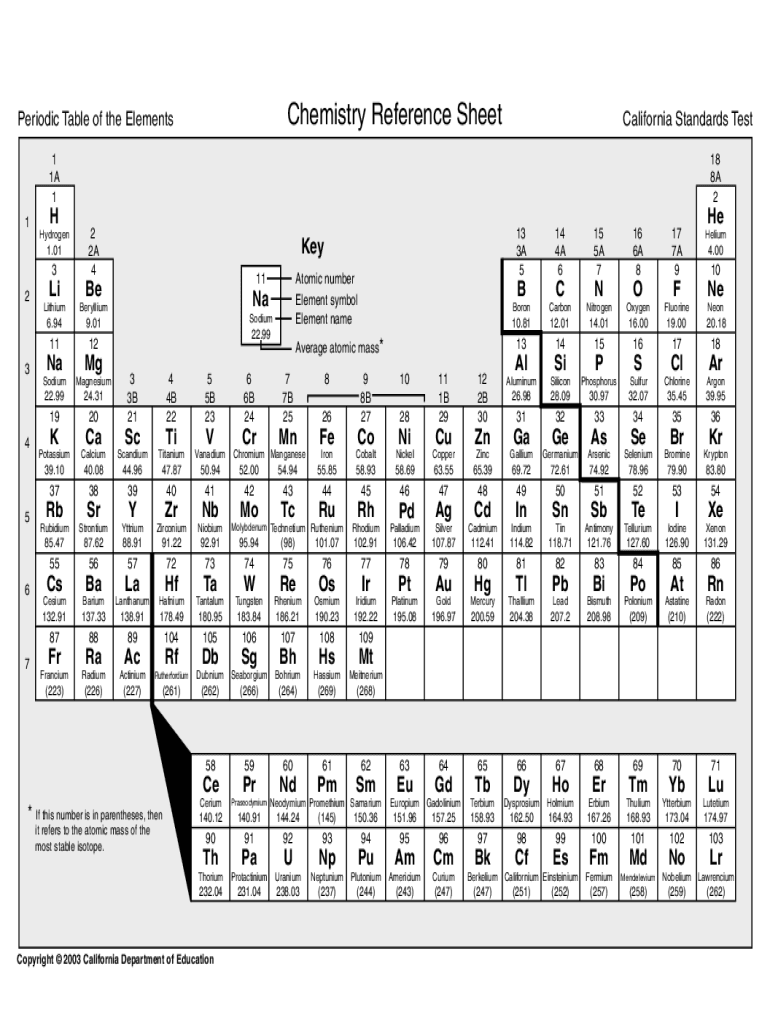 
g H 2O, must be used as a conversion factor to calculate the maximum amount of solute, ammonium iodide, NH 4I, that can dissolve in the given amount of solvent, 75.0 grams of water, H 2O.Chemistry Reference Table: An Essential Tool for Every Science Student IntroductionĬhemistry is an interesting and challenging topic that calls for in-depth knowledge and careful consideration of every nuance. In order to determine whether this solution is saturated or unsaturated, the solubility of ammonium iodide, NH 4I, which has a reported value of 172 g/100. Since the chemical formula for water, H 2O, is associated with the 100.-gram quantities in the denominators of the solubilities in Table 7.9.1, water, H 2O, is the solvent in this solution, and the remaining substance, ammonium iodide, NH 4I, is the solute, "by default." Use the solubility information that is presented in Table 7.9.1 to determine whether the resultant solution is saturated or unsaturated, and calculate the amount of excess solute that remains undissolved in this solution.īefore a solubility limit can be applied as a conversion factor, each substance that is referenced in the given problem must first be classified as a solute or a solvent. 
\)Ī solution is prepared by mixing 129 grams of ammonium iodide and 75.0 grams of water at 20 degrees Celsius.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
